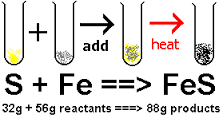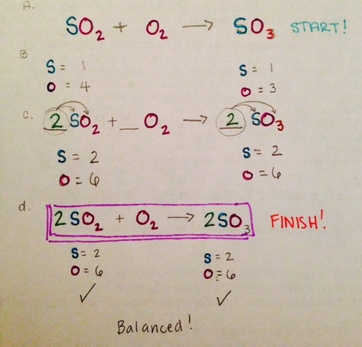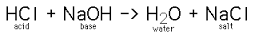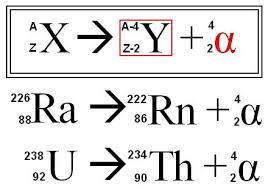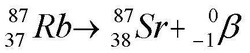The following list takes the Physical Science Standard and defines, decribes, pictures, links and/or explains the topics outlined in the course. This list is derived directly from the standards so it will contain most of the material you need to know for the exam.
PSc.2.1 Understand types, properties, and structure of matter.
Pure Substance- type of matter with a fixed composition.
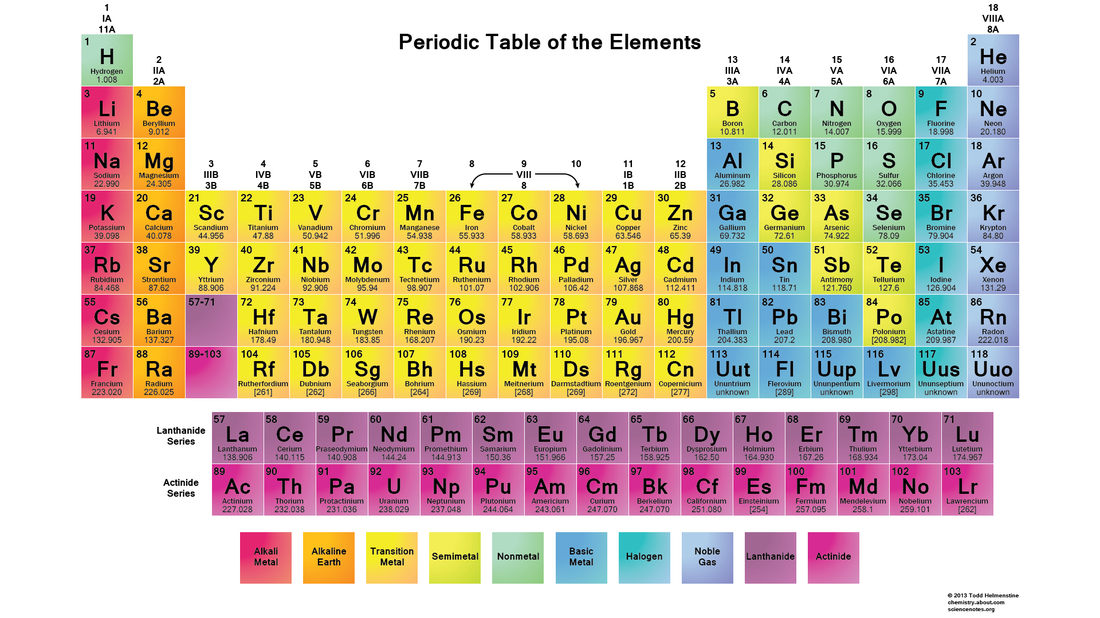
Elements made up of only one type of atom. Listed on the periodic table
Compounds- 2 or more different elements which are chemically combined
Homogeneous mixture physical combining of different elements and/or compounds taht are evenly mixed throughout
akasolution cannot be filtered does not scatter light will not settle out ex cola
examples : sugar water, salt water, soft drinks, filtered coffee/tea,
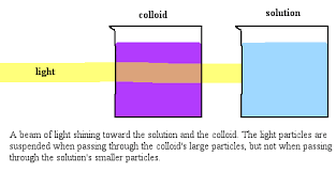
Colloid generally a homogeneous mixture cannot be filtered DOES SCATTER LIGHT will not settle out
Tyndall Effect
examples are fog, smoke, dust, whipped cream, shaving cream, marshmallows, jello, milk, mayonnaise, shampoo, et.al
Heterogeneous Mixture physical combining of elements and/or compounds that are not evenly mixed example - tossed salad, iced drink, cement,
Suspension can be filtered scatters light does settle particles visible
tossed salad, pizza, granite. drink wiht ice or pulp
Pure Substance- type of matter with a fixed composition.
Elements made up of only one type of atom. Listed on the periodic table
Compounds- 2 or more different elements which are chemically combined
Homogeneous mixture physical combining of different elements and/or compounds taht are evenly mixed throughout
akasolution cannot be filtered does not scatter light will not settle out ex cola
examples : sugar water, salt water, soft drinks, filtered coffee/tea,
Colloid generally a homogeneous mixture cannot be filtered DOES SCATTER LIGHT will not settle out
Tyndall Effect
examples are fog, smoke, dust, whipped cream, shaving cream, marshmallows, jello, milk, mayonnaise, shampoo, et.al
Heterogeneous Mixture physical combining of elements and/or compounds that are not evenly mixed example - tossed salad, iced drink, cement,
Suspension can be filtered scatters light does settle particles visible
tossed salad, pizza, granite. drink wiht ice or pulp
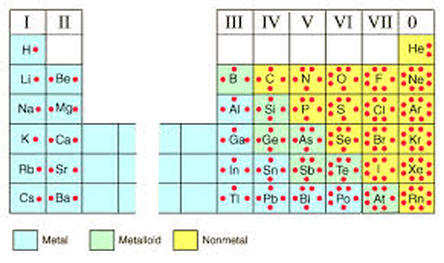
Classify an element as a metal, nonmetal, or metalloid based on its location on the periodic table.
Nonmetals
Located on the right side of the periodic table
Column 14 carbon 4 valence electrons oxidation number = +/- 4
Column 15 nitrogen, phosphorous 5 valence electrons oxidation number = -3
Column 16 oxygen, sulfur, selenium 6 valence electrons oxidation number = -2
Column 17 fluorine, chlorine, bromine, iodine, astatine 7 valence electrons oxidation number = -1
most reactive non-metal group known as the halogen group
Column 18, helium, neon, argon, krypton, xenon, radon 8 valence electrons oxidation number = 0
He only has 2 valence electrons but it is full.
Noble gas family all are gases - do not react with other elements (in normal circumstances)
Metalloids boron (B), silicon (Si), germanium (Ge), arsenic (As), antimony (Sb), tellurium (Te), polonium (Po) and astatine (At) are the elements found along the step like line between metals and non-metals of the periodic table
Characteristics
Share characteristics of both metals and non-metals and are also called semimetals. Metalloids are typically semi-conductors, which means that they both insulate and conduct electricity. This semi-conducting property makes Metalloids very useful as a computer chip material. Examples of metalloid elements are Silicon and Boron. (cliff's notes.com)
Metals are located to the left of thestairstep on the periodic table. They give away their valence electrons when combining with nonmetals and negative polyatomic ions
generally shiny, malleable (can be hammered into sheeets), ductile (can be pulled into wire), hard. good conductors of electricity. Examples of metals are Gold, Silver, Iron, Uranium, and Zinc.
Located on the right side of the periodic table
Column 14 carbon 4 valence electrons oxidation number = +/- 4
Column 15 nitrogen, phosphorous 5 valence electrons oxidation number = -3
Column 16 oxygen, sulfur, selenium 6 valence electrons oxidation number = -2
Column 17 fluorine, chlorine, bromine, iodine, astatine 7 valence electrons oxidation number = -1
most reactive non-metal group known as the halogen group
Column 18, helium, neon, argon, krypton, xenon, radon 8 valence electrons oxidation number = 0
He only has 2 valence electrons but it is full.
Noble gas family all are gases - do not react with other elements (in normal circumstances)
- Poor electrical conductors Brittle solids Little or no metallic luster Gain electrons easily
Metalloids boron (B), silicon (Si), germanium (Ge), arsenic (As), antimony (Sb), tellurium (Te), polonium (Po) and astatine (At) are the elements found along the step like line between metals and non-metals of the periodic table
Characteristics
Share characteristics of both metals and non-metals and are also called semimetals. Metalloids are typically semi-conductors, which means that they both insulate and conduct electricity. This semi-conducting property makes Metalloids very useful as a computer chip material. Examples of metalloid elements are Silicon and Boron. (cliff's notes.com)
Metals are located to the left of thestairstep on the periodic table. They give away their valence electrons when combining with nonmetals and negative polyatomic ions
generally shiny, malleable (can be hammered into sheeets), ductile (can be pulled into wire), hard. good conductors of electricity. Examples of metals are Gold, Silver, Iron, Uranium, and Zinc.
STATES OF MATTER
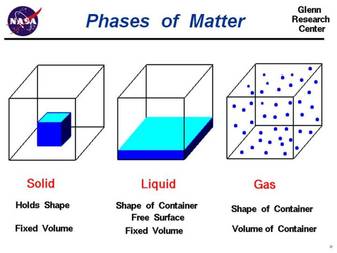
Solid definite volume does not compress definite shape
Liquid definite volume does not compress takes the shape of the container
Gas no definite volume can compress takes the shape of the container
Change of state of matter (physical property and physical change)
Solid to Liquid = melting Liquid to gas = evaporation Solid to Gas (no liquid phase) = sublimation (dry ice )
Liquid to solid = freezing Gas to Liquid = condensation Gas to Solid (no liquid phase) = deposition (snow forms this way)
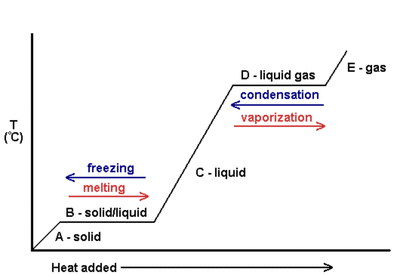
PHASE CHANGE DIAGRAM be able to read and interpret for:
melting point/freezingpoint (lowest flat spot) contains solid and liquid during the phase change
point of vaporization/condensation point (highest flat spot) contains liquid and gas during the phase change
solid only (lowest diagonal), liquid only (middle diagonal), gas only (upper diagonal)
Solid definite volume does not compress definite shape
Liquid definite volume does not compress takes the shape of the container
Gas no definite volume can compress takes the shape of the container
Change of state of matter (physical property and physical change)
Solid to Liquid = melting Liquid to gas = evaporation Solid to Gas (no liquid phase) = sublimation (dry ice )
Liquid to solid = freezing Gas to Liquid = condensation Gas to Solid (no liquid phase) = deposition (snow forms this way)
PHASE CHANGE DIAGRAM be able to read and interpret for:
melting point/freezingpoint (lowest flat spot) contains solid and liquid during the phase change
point of vaporization/condensation point (highest flat spot) contains liquid and gas during the phase change
solid only (lowest diagonal), liquid only (middle diagonal), gas only (upper diagonal)
Solvents and solutes when a simple solution is basically two substances that are evenly mixed together.the
solute, usually the lesser amount is the substance to be dissolved (sugar).
The solvent is the one doing the dissolving (water). Water (a polar molecule) is often called the UNIVERSAL SOLVENT
LIKES DISSOLVE LIKES polar dissolves polar, nonpolar dissolves nonpolar
BUT polar does not dissolve nonpolar i.e. water(polar) does NOT dissolve grease (non-polar)
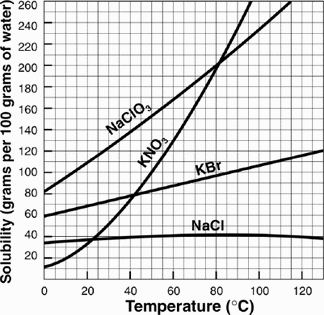
Saturated solution that is unable to absorb or dissolve any more of a solute at a given temperature and pressure. FULL
Unsaturated solution that is able to absorb or dissolve any more of a solute at a given temperature and pressure. Not FULL
Supersaturated contains more solute, or dissolved material, than it would under normal conditions
Solubility curve given a temperature go vertical until you touch the line of the solute, go horizontal to determine tha amount that is dissolved. This point determines the saturation point. If given a temperature and it is below the solute's curve it is unsaturated, if the amount is above the line it is supersaturated.
solute, usually the lesser amount is the substance to be dissolved (sugar).
The solvent is the one doing the dissolving (water). Water (a polar molecule) is often called the UNIVERSAL SOLVENT
LIKES DISSOLVE LIKES polar dissolves polar, nonpolar dissolves nonpolar
BUT polar does not dissolve nonpolar i.e. water(polar) does NOT dissolve grease (non-polar)
Saturated solution that is unable to absorb or dissolve any more of a solute at a given temperature and pressure. FULL
Unsaturated solution that is able to absorb or dissolve any more of a solute at a given temperature and pressure. Not FULL
Supersaturated contains more solute, or dissolved material, than it would under normal conditions
Solubility curve given a temperature go vertical until you touch the line of the solute, go horizontal to determine tha amount that is dissolved. This point determines the saturation point. If given a temperature and it is below the solute's curve it is unsaturated, if the amount is above the line it is supersaturated.
Physical Properties vs Chemical Properties
Physical Properties of Metals: Hardness, Ductility, Melting Point, etc.
Physical Properties of Nonmetals: Dull, Low Density, Low Melting Point, etc.
Physical Properties of Metalloids: Ductile, Malleable, etc.
Physical property terms: color, smell. freezing point, boiling point, melting point, viscosity, magnetic, density
STATES OF MATTER FOR ALL Elements, Compounds, Mixtures are physical properties
Density: (formula is on reference table) The density of a substance is the relationship between the mass of the substance and how much space it takes up (volume). Formula Density = mass/volume D=m/v unit is g/cm3
Volume of liquid is ml 1 cm3 = 1ml
water displacement calculation; Subtract the original amount from the measurement in a graduated clylinder from the new volume after an object has been placed into it.
Chemical Properties of Metals: Corrosion, Form Positive Ions, etc.
Chemical Properties of Nonmetals: React with metals, Gain Valence Electrons, etc.
Chemical Properties of Metalloids: To the right of the stair step on the periodic table the elements gain electrons when bonding and to the left of the stair step, the elements lose electrons when bonding.
Physical change- no new material is formed
Chemical change- a new material is formed and you are unable to get the original material back examples: burnt toast, rusting nails, all chemical reactions 2Na + Cl2 -> 2NaCl, precipitate forms, effervesence (bubbles formed),
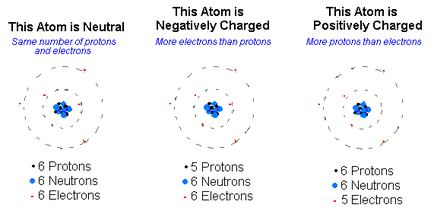
Describe the charge, relative mass, and the location of protons, electrons, and neutrons within an atom.
Location Charge charge relative mass (atomic mass units)
proton Inside the nucleus Positive p+ 1amu
electron On the rings(regions) Negative e- 0 "no mass"
outside of the nucleus
neutron Inside the nucleus Neutral n0 1 amu
Calculate the number of protons, neutrons, electrons, and mass number in neutral atoms and ions
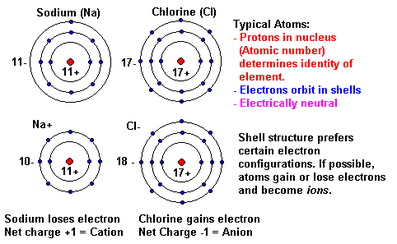
Atomic numberof an element = the number of protons example Sodium (Na) is element #11 and it has 11p+
In a neutral atom # protons = # elctrons
To calculate neutrons: round the atomic mass number and subtract the atomoic number (protons)
Example Sodium (Na) has an attomic mass of 22.99 round up to( 23) subract the atomic number (11) = 12 neutrons
Ions - electrically charged atoms due to gaining electrons (becomes a negative ion - anion or losing electrons (becomes a positive ions - cation)
Physical Properties of Metals: Hardness, Ductility, Melting Point, etc.
Physical Properties of Nonmetals: Dull, Low Density, Low Melting Point, etc.
Physical Properties of Metalloids: Ductile, Malleable, etc.
Physical property terms: color, smell. freezing point, boiling point, melting point, viscosity, magnetic, density
STATES OF MATTER FOR ALL Elements, Compounds, Mixtures are physical properties
Density: (formula is on reference table) The density of a substance is the relationship between the mass of the substance and how much space it takes up (volume). Formula Density = mass/volume D=m/v unit is g/cm3
Volume of liquid is ml 1 cm3 = 1ml
water displacement calculation; Subtract the original amount from the measurement in a graduated clylinder from the new volume after an object has been placed into it.
Chemical Properties of Metals: Corrosion, Form Positive Ions, etc.
Chemical Properties of Nonmetals: React with metals, Gain Valence Electrons, etc.
Chemical Properties of Metalloids: To the right of the stair step on the periodic table the elements gain electrons when bonding and to the left of the stair step, the elements lose electrons when bonding.
Physical change- no new material is formed
Chemical change- a new material is formed and you are unable to get the original material back examples: burnt toast, rusting nails, all chemical reactions 2Na + Cl2 -> 2NaCl, precipitate forms, effervesence (bubbles formed),
Describe the charge, relative mass, and the location of protons, electrons, and neutrons within an atom.
Location Charge charge relative mass (atomic mass units)
proton Inside the nucleus Positive p+ 1amu
electron On the rings(regions) Negative e- 0 "no mass"
outside of the nucleus
neutron Inside the nucleus Neutral n0 1 amu
Calculate the number of protons, neutrons, electrons, and mass number in neutral atoms and ions
Atomic numberof an element = the number of protons example Sodium (Na) is element #11 and it has 11p+
In a neutral atom # protons = # elctrons
To calculate neutrons: round the atomic mass number and subtract the atomoic number (protons)
Example Sodium (Na) has an attomic mass of 22.99 round up to( 23) subract the atomic number (11) = 12 neutrons
Ions - electrically charged atoms due to gaining electrons (becomes a negative ion - anion or losing electrons (becomes a positive ions - cation)
Explain how the different mass numbers of isotopes contributes to the average atomic mass for a given element (conceptual, no calculations).
Isotope different versions of the same element and in each isotope the number of protons always stays the same while the number of neutrons varies.
Use isotopic notation to write symbols for various isotopes (ex. Carbon-12, C-12, 12C, etc.)
Each of the examples in the goal above indicate that the atom is carbon, the atomic mass is 12 Each atom would have 6 protons, 6 electrons and 6 neutrons
If you had C-14 you still have carbon with 6 p+, 6- and 8 neutrons
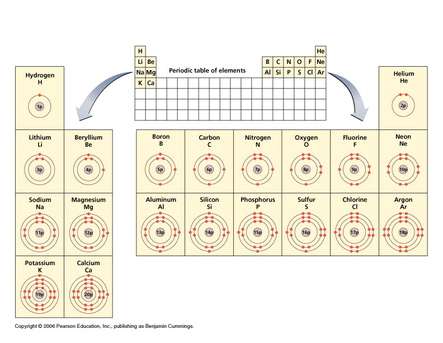
Explain Bohr’s model of the atom
Protons and neutrons will be located in the nucleus (must = atomic mass)
1st shell = max 2 electrons
2nd shell = max 8 electrons
3rd shell = max 8 electrons
Isotope different versions of the same element and in each isotope the number of protons always stays the same while the number of neutrons varies.
Use isotopic notation to write symbols for various isotopes (ex. Carbon-12, C-12, 12C, etc.)
Each of the examples in the goal above indicate that the atom is carbon, the atomic mass is 12 Each atom would have 6 protons, 6 electrons and 6 neutrons
If you had C-14 you still have carbon with 6 p+, 6- and 8 neutrons
Explain Bohr’s model of the atom
Protons and neutrons will be located in the nucleus (must = atomic mass)
1st shell = max 2 electrons
2nd shell = max 8 electrons
3rd shell = max 8 electrons
Lewis Dot Structures show the element symbol and the valence electrons only (outer shell electrons)
PSc.2.2 Understand chemical bonding and chemical interactions
Infer valence electrons, oxidation number, and reactivity of an element based on its location in the Periodic Table
Valence electrons = the number of electrons in the outer shell See Lewis dot structure above
Oxidation numbers positive or negative number that indicates the # of electrons an atom gains, loses or shares to become stable
Periodic Table write these numbers above the appropriate group on the periodic table on your refernce tabel
Group 1 - Alkali Metals 1 valence e- oxidation number = +1
Group 2 Alkaline Earth Metals 2 valence e- oxidation number = +2
Transition Metals (groups 3-12 valence & oxidations may vary idicated by the name + roman numeral for the oxidation number i.e.
Iron has two osidation # Iron(II) = Fe 2+ and Iron (III) Fe 3+
Group 13 Boron Family 3 valence e- oxidation number = +3
Group 14 Carbon Family 4 valence e- oxidation number = + or - 4
Group 15 Nitrogen Family 5 valence e- oxidation number = - 3
Group 16 Oxygen Family 6 valence e- oxidation number = -2
Group 17 HALOGENS 7 valence e- oxidation number = -1
Group 18 Noble Gases 8 valence e- oxidation number = 0 Shell is full - no need to reacte He is full with 2e- on 1st shell
When they are called a compound (ionic bonds ) or molecules (covalent bonds). These are binary compounds.
Three or more elements combining together may have polyatomic ions in them.
Polyatomic ions two or more atoms covalently bonded or of a metal complex that can be considered to be acting as a single unit.
Each type of bond has specific rules for naming them (nomenclature)
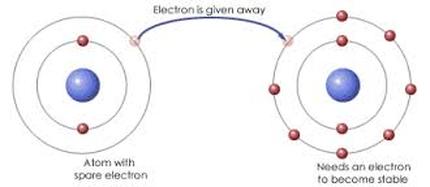
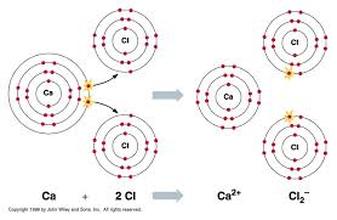
Ionic Bonding when a positively charged ion forms a bond with a negatively charged ions and one atom transfers electrons to another. Metal + Nonmetal
Infer valence electrons, oxidation number, and reactivity of an element based on its location in the Periodic Table
Valence electrons = the number of electrons in the outer shell See Lewis dot structure above
Oxidation numbers positive or negative number that indicates the # of electrons an atom gains, loses or shares to become stable
Periodic Table write these numbers above the appropriate group on the periodic table on your refernce tabel
Group 1 - Alkali Metals 1 valence e- oxidation number = +1
Group 2 Alkaline Earth Metals 2 valence e- oxidation number = +2
Transition Metals (groups 3-12 valence & oxidations may vary idicated by the name + roman numeral for the oxidation number i.e.
Iron has two osidation # Iron(II) = Fe 2+ and Iron (III) Fe 3+
Group 13 Boron Family 3 valence e- oxidation number = +3
Group 14 Carbon Family 4 valence e- oxidation number = + or - 4
Group 15 Nitrogen Family 5 valence e- oxidation number = - 3
Group 16 Oxygen Family 6 valence e- oxidation number = -2
Group 17 HALOGENS 7 valence e- oxidation number = -1
Group 18 Noble Gases 8 valence e- oxidation number = 0 Shell is full - no need to reacte He is full with 2e- on 1st shell
When they are called a compound (ionic bonds ) or molecules (covalent bonds). These are binary compounds.
Three or more elements combining together may have polyatomic ions in them.
Polyatomic ions two or more atoms covalently bonded or of a metal complex that can be considered to be acting as a single unit.
Each type of bond has specific rules for naming them (nomenclature)
Ionic Bonding when a positively charged ion forms a bond with a negatively charged ions and one atom transfers electrons to another. Metal + Nonmetal
Naming ionic compounds
Always write the symbol for the metal first then the nonmetal
Write the oxidation number above each element.
If the oxidation number = 0 when added together then it is a stable and neutral compound
If the oxidation #s do not equal 0, then use the criss-cross method
Then name the compound the metal keeps its name and the nonmetal drops the ending and adds -ide
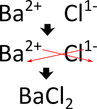
Barium + Chlorine transitions use Roman numerals Iron 3+ and Oxygen 2-
barium chloride Iron (III) Oxide
Always write the symbol for the metal first then the nonmetal
Write the oxidation number above each element.
If the oxidation number = 0 when added together then it is a stable and neutral compound
If the oxidation #s do not equal 0, then use the criss-cross method
Then name the compound the metal keeps its name and the nonmetal drops the ending and adds -ide
Barium + Chlorine transitions use Roman numerals Iron 3+ and Oxygen 2-
barium chloride Iron (III) Oxide
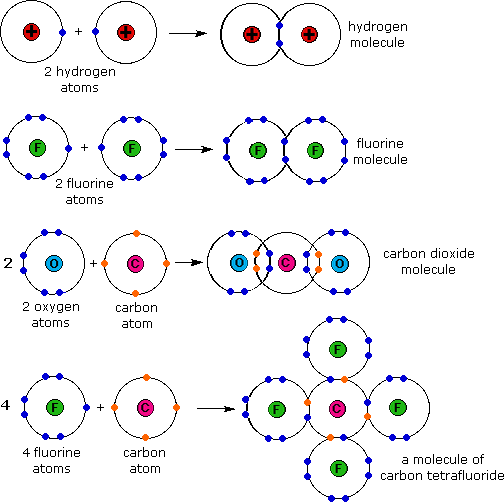
Covalent Bonding involves the sharing of electron pairs between atoms Nonmetal + Nonmetal
Naming covalent bonds: Make sure the elements are all nonmetals
the first element keeps it name and the 2nd drops the ending and adds -ide.
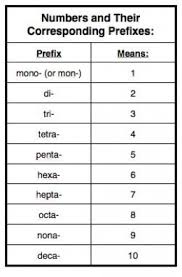
For Covalent you will use the prfixes to tell how many atoms are present In most moluecules you will not mono- before the first word
the first element keeps it name and the 2nd drops the ending and adds -ide.
For Covalent you will use the prfixes to tell how many atoms are present In most moluecules you will not mono- before the first word
Metallic Bonding is the bonding between molecules within metals called alkali reactive force. It is the sharing of a sea of delocalised electrons amongst a lattice of positive ions, where the electrons act as a "glue" giving the substance a definite structure. Metal + Metal
Predict chemical formulas and names for simple compounds based on knowledge of bond formation and naming conventions
Writing chemical formulas
1.The cation is always named first and the anion second.
2. A simple cation (obtained from a single atom) takes its name from the name of the element.
3. A simple anion (obtained from a single atom) is named by taking the first part of the element name (the root) and adding the letters “IDE.”
4. Write the name for the compound by combining the names of the ions.
Writing chemical formulas
1.The cation is always named first and the anion second.
2. A simple cation (obtained from a single atom) takes its name from the name of the element.
3. A simple anion (obtained from a single atom) is named by taking the first part of the element name (the root) and adding the letters “IDE.”
4. Write the name for the compound by combining the names of the ions.
Polyatomic Ions Compounds whuch are charged ions composed of two or more atoms covalently bonded.
Ammonium ion NH4+ is the only positive PI you need to know it acts like a metal and will give away the extra electron. OH- (hydroxide) needs to gain one electron.
Put them together and you have NH4OH = ammonium hydroxide
USE A CHART or REFERENCE TABLE TO GIVE YOU THE NAME, FORMIula, and OXIDATION MUMBER
Ammonium ion NH4+ is the only positive PI you need to know it acts like a metal and will give away the extra electron. OH- (hydroxide) needs to gain one electron.
Put them together and you have NH4OH = ammonium hydroxide
USE A CHART or REFERENCE TABLE TO GIVE YOU THE NAME, FORMIula, and OXIDATION MUMBER
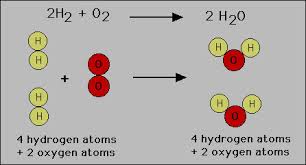
PSc.2.2.4 Exemplify the law of conservation of mass by balancing chemical equations.
The law of conservation of matter, matter is neither created nor destroyed, so we must have the same number and type of atoms after the chemical change as were present before the chemical change.
The law of conservation of matter, matter is neither created nor destroyed, so we must have the same number and type of atoms after the chemical change as were present before the chemical change.
Balancing Equation a chemical equation in which there are equal numbers of each type ofatom on each side of the equation
(see above)
Coefficient a multiplier that must go in front of all compounds to balance the number of each type of element on both sides of the euation.
(see above)
Coefficient a multiplier that must go in front of all compounds to balance the number of each type of element on both sides of the euation.
PSc.2.2.5 Classify types of reactions such as synthesis, decomposition, single replacement, or double replacement
All equations have two sides the left side are the REACTANTS -> right side are the PRODUCTS
5 basic types of equations for physical science Note: combination is what we called synthesis
All equations have two sides the left side are the REACTANTS -> right side are the PRODUCTS
5 basic types of equations for physical science Note: combination is what we called synthesis
PSc.2.2.6 Summarize the characteristics and interactions of acids and bases.
Compounds that add H+ ions to water when in a solution. Compouns start with hydrogen (not are acidic all compound that do start with the H. ( pere water, H2O is not acidic)
Compounds that add H+ ions to water when in a solution. Compouns start with hydrogen (not are acidic all compound that do start with the H. ( pere water, H2O is not acidic)
ACIDS produce the H+ ion in solution BASES produce the OH- ion in solution
Examples: HCl - hydrochloric acid NaOH - sodium hydroxide
H2SO4 - sulfuric acid Al(OH)3 aluminum hydroxide
Examples: HCl - hydrochloric acid NaOH - sodium hydroxide
H2SO4 - sulfuric acid Al(OH)3 aluminum hydroxide
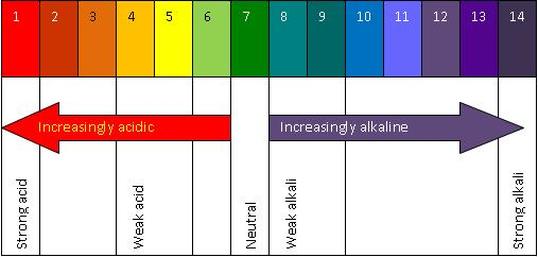
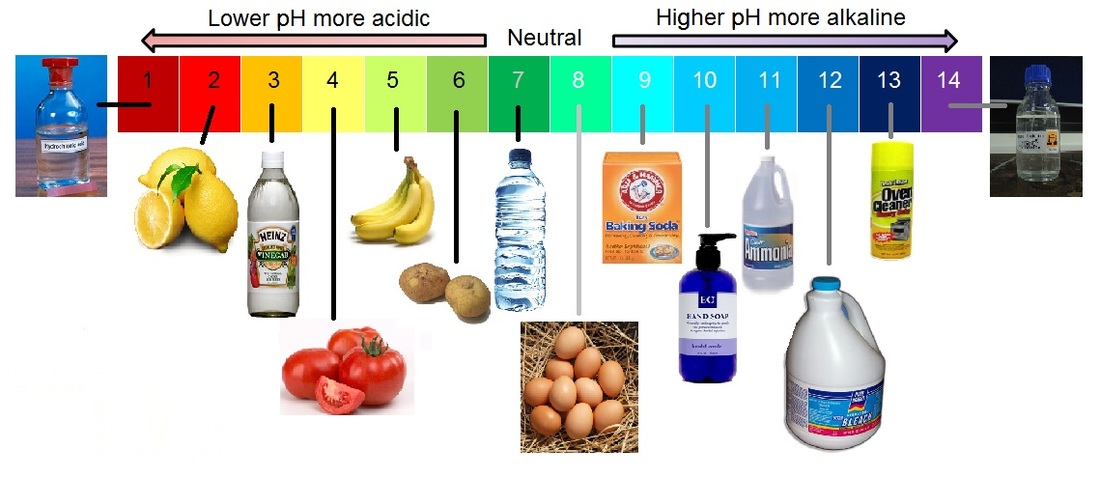
pH Scale In chemistry, pH (/piːˈeɪtʃ/) is the negative log of the activity of the hydrogen ion in an aqueous solution.
pH less than 7 are said to be acidic and solutions with a pH greater than 7 are basic or alkaline
Pure water has a pH of 7
pH less than 7 are said to be acidic and solutions with a pH greater than 7 are basic or alkaline
Pure water has a pH of 7
Acids + Bases reactions (neurtalization reaction)
Forms a double replacement reation and the products will be a salt and water.
Forms a double replacement reation and the products will be a salt and water.
PSc.2.3 Understand the role of the nucleus in radiation and radioactivity.
PSc.2.3.1 Compare nuclear reactions including: alpha decay, beta decay, and gamma decay; nuclear fusion and nuclear fission.
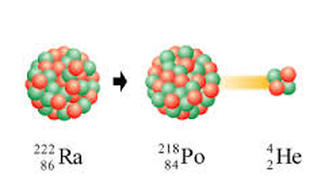
Alpha decay:
1) The nucleus of an atom splits into two parts.
2) One of these parts (the alpha particle) goes zooming oalpha can stopped byff into space.
3) The nucleus left behind has its atomic number reduced by 2 and its mass number is reduced by 4
* alpha particls can be stopped by a sheet of paper
PSc.2.3.1 Compare nuclear reactions including: alpha decay, beta decay, and gamma decay; nuclear fusion and nuclear fission.
Alpha decay:
1) The nucleus of an atom splits into two parts.
2) One of these parts (the alpha particle) goes zooming oalpha can stopped byff into space.
3) The nucleus left behind has its atomic number reduced by 2 and its mass number is reduced by 4
* alpha particls can be stopped by a sheet of paper
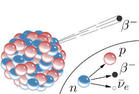
Beta Becay 1) A neutron inside the nucleus of an atom breaks down, changing into a proton.
2) It emits an electron and an anti-neutrino (more on this later) which go zooming off into space.
3) The atomic number goes UP by one and mass number remains unchanged.
beta particlees can be stopped by a sheet of aluminium foil
2) It emits an electron and an anti-neutrino (more on this later) which go zooming off into space.
3) The atomic number goes UP by one and mass number remains unchanged.
beta particlees can be stopped by a sheet of aluminium foil
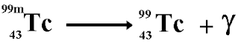
Gamma Decay composed ofelectromagnetic rays. It is distinguished from x-rays only by the fact that it comes from the nucleus.
No mass
gamma can be stopped by several cm of lead
No mass
gamma can be stopped by several cm of lead
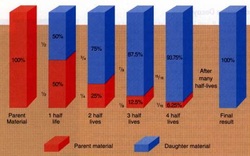
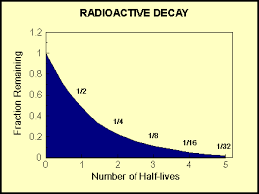
Half-Life: amount of time it takes for half the nuclei in a sample of a radioactive isotope to decay